
Get Care Now
If you’re in need of care, it’s important to know which service is the right one for your situation.
Primary Care
Walk-In Clinics
Urgent Care
Emergency Care
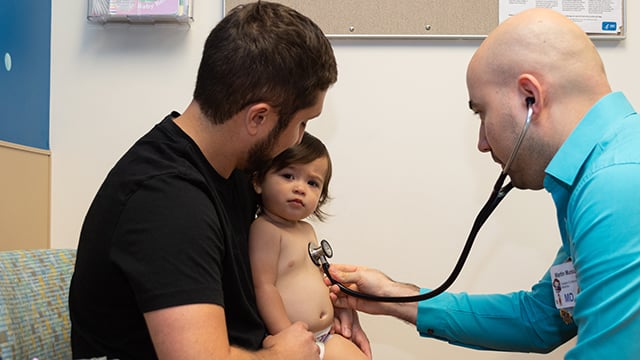
Primary Care
Our primary care physicians (PCPs) offer a wide range of preventive wellness, screening, and healthcare services, each tailored to your individual needs. A PCP is your main healthcare provider in non-emergency situations. Your PCP's role is to:
- Provide preventive care and teach healthy lifestyle choices
- Identify and treat common medical conditions
- Assess the urgency of your medical problems and direct you to the best place for care
- Make referrals to medical specialists when necessary

Walk-In Clinic
We have two walk-in clinics that are ready to serve you in those situations where you can’t wait for a visit to your primary care provider.
- Reid Ready Care Walk-in Clinic can handle many routine illnesses such as a sore throat, stuffy or runny nose, earache, and similar ailments.
- Reid Health Ortho Walk-in NOW specializes in treating unexpected injuries or nagging bone/joint pains.

Urgent Care
When you need prompt care but aren’t ill enough for a trip to the Emergency Room, our four urgent care facilities are here to help. Each can treat a wide variety of illnesses, conditions, and non-life-threatening injuries.
- Reid Health Urgent Care
- Reid Health Urgent Care – Connersville

Emergency Care
Our teams of emergency medicine providers, surgeons, and anesthesiologists in Richmond and Connersville are prepared to help you in times of serious and life-threatening conditions. We have been certified as a Primary Stroke Center, and our Richmond facility has been verified as a Level III Trauma Center.

Specialty Care
Our specialty care services bring advanced medicine and compassionate expertise together, close to home. Specialists work collaboratively with your other care providers to help coordinate diagnosis and treatment for the best care possible.
Need help scheduling an appointment?
Whether you’re a new patient or an existing one seeking a new provider, we’re here to help you find the care you need.